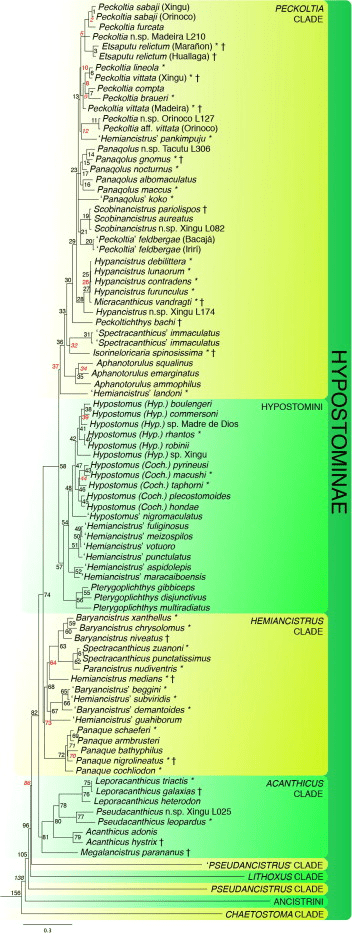
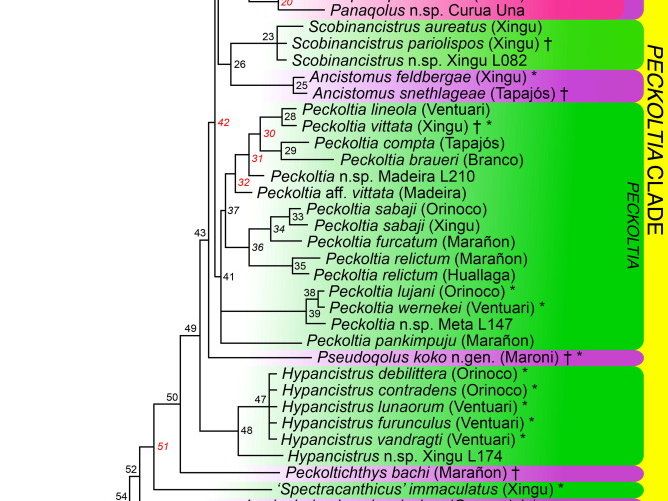
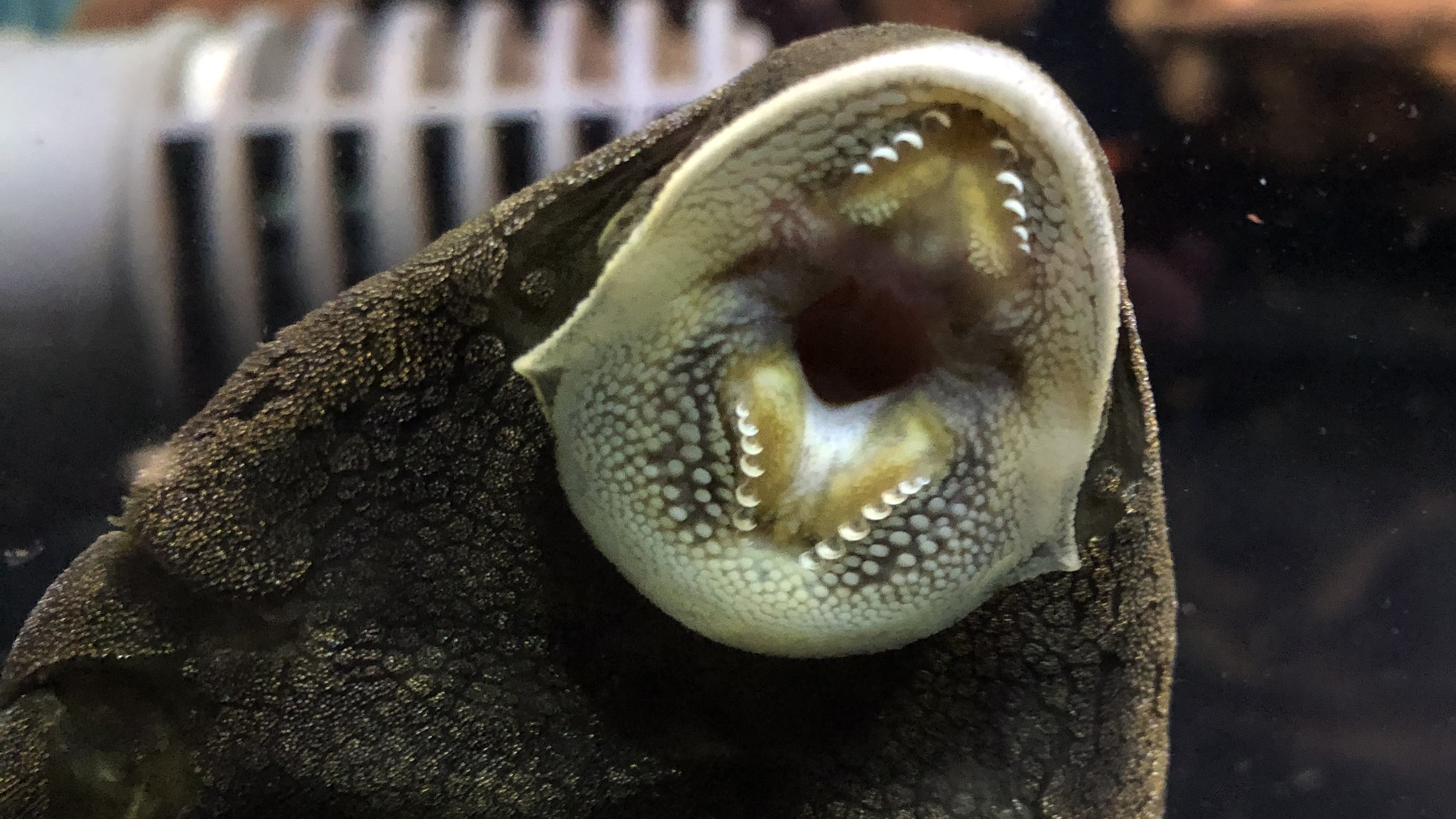
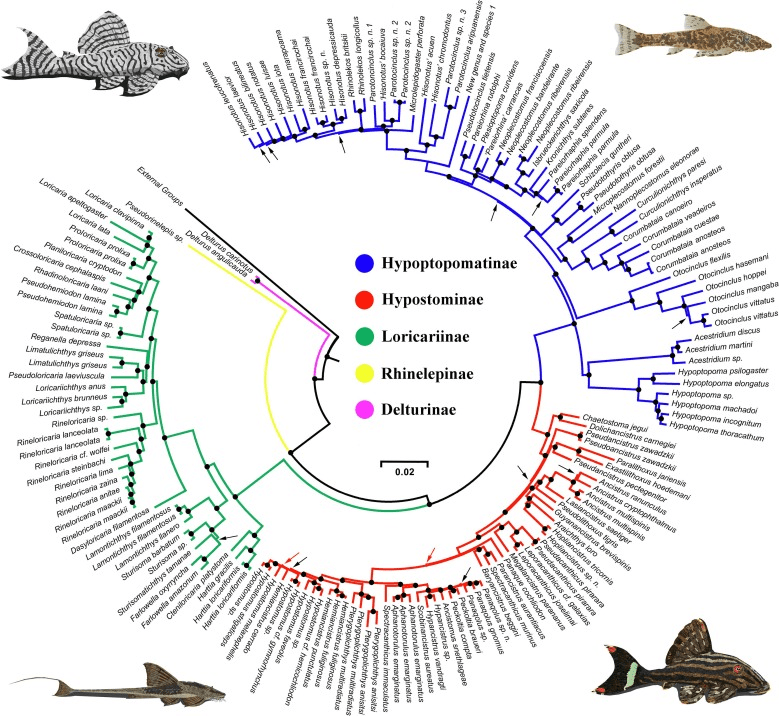
I am personally most charmed by those Loricariids with unusual anatomy and none more then that dorso-ventrally compressed body shape. Most of these fishes enjoy high velocity water, living in the cracks and crevices of the rocks present, more then often not fishes you’d find around plants or even wood. Two genera come to mind when we think of this, Chaetostoma and Ancistrus although many more do exploit such a niche like Pseudolithoxus. Many genera we do not see in the trade though, these are largely members of that Chaetostoma clade; the paraphyletic Cordylancistrus, Andeanancistrus, Transancistrus and Leptoancistrus (Lujan et al., 2015).

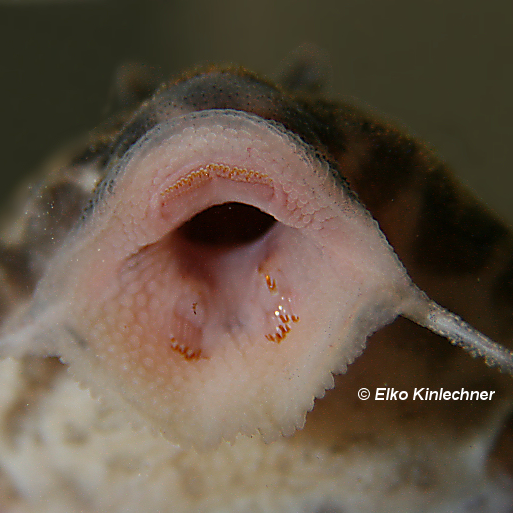
This Chaetostoma clade have a unique appearance of long wide jaws and dermal plating stopping before the end of the head leaving a fleshy rim that lacks tentacles. The only taxa that look similar would be a few Ancistrus and the Neoplecostominae, Pareiorhapis but the latter displays quite reasonable hypertrophied odontodes and a much wider head. There is more precise skeletal anatomy to identify Chaetostoma (Lujan et al., but not really the easiest for the fishkeeper to identify.
As of 2022 there were 49 currently described species in the genus (Meza-Vargas et al., 2022) making Chaetostoma one of the numerous Loricariid genera excluding Ancistrus and Hypostomus all of which have many undescribed species. While in the aquarium trade we see very few with some rarities appear on occasion, the majority seem to be imported rather generally and therefore bycatch is not rare. This is one of the genera you can find something almost unseen in a general fish store due to this lack of identification. The most common being Chaetostoma formosae, C. sp. ‘L147’ and C. dorsale but Chaetostoma sp. L455/L457 is not unseen and has some amazingly striking patterning.

Chaetostoma has often been associated with being small but this genus represents some larger species such as Chaetostoma brevilabiatum growing to over 18cm Standard Length (SL) although most are around that 6/7cm SL mark (Lujan et al., 2015). They are notorious fast growers if in the right setup so certainly not one to forget about upgrading soon enough.
Habitat
While largely a hillstream or high velocity fish (In terms of our fishkeeping) they can be particularly widespread or less so depending on the species (Lujan et al., 2015). Ecology is rarely recorded as with many fishes where taxonomy has been the focus and while most descriptions come with coordinates that can be crosschecked against other information it is still somewhat making assumptions. Although for Chaetostoma chimu, C. formosae, C. dorsale, C. platyrhynchus and C. joropo all three are found in the same locality, explains mixed imports and we do have ecological records focused on C. chimu. The water is well oxygenated and by our aquarium standards has a high flow, temperatures of 21-29c, a pH of 7.1-8.9 along with a conductivity of 20.4–269.0 μS (Urbano‐Bonilla & Ballen, 2021) . This suggests fishes that experience quite a bit of variation and swings seasonally or maybe if there is frequent rainfall. Generally this does infer maybe a few species not difficult to house in captivity. That neutral pH is not uncommonly recorded, Chaetostoma spondylus is recorded from a habitat of a pH of 7.1 with again highly oxygenated water (Salcedo & Ortega 2015). Chaetostoma joropo also inhabiting highly oxygenated water at a pH of 7.1-8.6, a conductivity of 10.4–258.0 μS and temperatures of 21-30c. These are certainly not fish to keep at least at the high extreme but given the locality of many it seems particularly those at higher elevations would need much cooler water year round.

These habitats are extremely rocky with round boulders weathered from the flow of the rivers (Urbano‐Bonilla& Ballen, 2021; Meza-Vargas et al., 2022). Whether it be rocks, wood, pleco caves etc. plenty of hiding spots are a must for this genus.
Diet
While the habitat of Chaetostoma proves them adaptable their diet might not, these elongate jaws are extremely similar to other genera that have provided a challenge to aquarists e.g. Baryancistrus. The longer jaws with more numerous teeth are strongly associated with algivory (feeds on mostly algaes/aufwuch/periplankton; Lujan et al., 2012). Zúñiga-Upegui et al. (2017) is probably the most detailed paper on the diet of Chaetostoma although few ever discuss their diet, from their analysis the genus feeds almost entirely on algaes particularly diatoms. These diatoms are unlikely to be those highly stubborn ones to cause issues in the aquarium though.
If anything much like Baryancistrus this is a genus who would benefit from large amounts of algae’s in their diet whether it be Repashy soilent green with additional algal powders mixed in or In The Bag’s Pleco Pops. Many fish diets even most claimed as algae wafers contain very little and this genus has shown adaptable to these diets nutritionally I can’t see them being ideal.
Behaviour
I can’t argue for or against their territoriality as I haven’t seen it, even with any territorial species there is a benefit in others for enrichment given the right amount of space. Many fishes seem to learn feeding behaviours off each other.

Conclusion
Chaetostoma, it’s so unusual looking and can look creepy, they often get forgotten. These algivores who enjoy high flow and velocity would certainly make interesting tankmates in some of those aquariums which allow for such seasonal variation. Adaptable in parameters and maybe less so in diet is probably what defines the genus.
References/species descriptions:
Lujan, N. K., Meza-Vargas, V., Astudillo-Clavijo, V., Barriga-Salazar, R., & López-Fernández, H. (2015). A multilocus molecular phylogeny for Chaetostoma clade genera and species with a review of Chaetostoma (Siluriformes: Loricariidae) from the Central Andes. Copeia, 103(3), 664-701.
Lujan, N. K., Winemiller, K. O., & Armbruster, J. W. (2012). Trophic diversity in the evolution and community assembly of loricariid catfishes. BMC Evolutionary Biology, 12(1), 1-13.
Meza-Vargas, V., Calegari, B. B., Lujan, N. K., Ballen, G. A., Oyakawa, O. T., Sousa, L. M., … & Reis, R. E. (2022). A New Species of Chaetostoma (Siluriformes: Loricariidae) Expands the Distribution of Rubbernose Plecos Eastward into the Lower Amazon Basin of Brazil. Ichthyology & herpetology, 110(2), 364-377.
Salcedo, N. J., & Ortega, H. (2015). A new species of Chaetostoma, an armored catfish (Siluriformes: Loricariidae), from the río Marañón drainage, Amazon basin, Peru. Neotropical Ichthyology, 13, 151-156.
Urbano‐Bonilla, A., & Ballen, G. A. (2021). A new species of Chaetostoma (Siluriformes: Loricariidae) from the Orinoco basin with comments on Amazonian species of the genus in Colombia. Journal of Fish Biology, 98(4), 1091-1104.
Zúñiga-Upegui, P. T., Villa-Navarro, F. A., García-Melo, L. J., García-Melo, J. E., Reinoso-Flórez, G., Gualtero-Leal, D. M., & Ángel-Rojas, V. J. (2014). Aspectos ecológicos de< em> Chaetostoma sp.(Siluriformes: Loricariidae) en el alto río Magdalena, Colombia. Biota Colombiana, 15(2).