Natural has been the trigger word for the aquarium hobby for a long time, it’s a great way to sell a concept or product. You’ll commonly see claims like natural is more stable, healthier or the most extreme something along the lines of the only way. The word natural sells (Scott et al., 2020) and many of us get an aquarium to replicate a pocket of the natural world in our own home. But is it really natural, and is natural good?
Vitamin Green
Humans have a natural tendency to be attracted to nature and this was coined by the theory known as biophilia (Wilson, 2007) and there are many potential benefits of humans interacting with the natural world (2017). This is a very human centric way to think about the world, thinking about the world and how it benefits us but not what else is around us.

What we see as natural?
There seems to be no strict definition for natural and it definitely seems to be up to ones own interpretation. Our perception of nature is massively distorted, not much of the world is unchanged by humans and nothing more then freshwater (Eastwood et al., 2023). Our influence on freshwater has massively changed how freshwater ecosystems function and the species present (Søndergaard & Jeppesen, 2007). One of these changes has been the introduction of invasive species and although we commonly associate animals with this, plants can be invasive too. Invasive plant species can effect how rivers and lakes function along with the species present (Schultz & Dibble, 2012).
When we go out to see our local water ways, the anthropogenic influence is easily dismissed as it has always been there. American skunk cabbage as an invasive species to the UK was first sighted in 1947 (Department for Environmental and Rural Affairs et al., 2019). Elodea spp. on the other hand has been invasive in the UK since 1879 reproducing vegetatively due to a lack of male plants (Simpson, 1986).
Availability Bias
Of course when I talk about what we see locally as being natural or anthropogenic I am often referring to North American and European ecosystems which differ a lot from many popular locations we obtain fishes from. Most of us aren’t keeping native fishes so to understand natural we should think mostly about the ecosystems of South America, South East Asia and Africa. Here comes with the issue of availability bias, where information might be sparse for certain habitats.

Most fishkeepers are not looking at scientific papers which is obviously a lot more diverse so here we have to look at social media, websites and Youtube. Here we are confronted largely by ecosystems dense in vegetation, when it is not it is black water with leaves. While botanicals have been used to almost fight against the planted idea of nature it has come at the cost of many believing this is what represents freshwater in the tropics. Here comes the bias, if this is what we see is this all that there is?
Many freshwater ecosystems might not be accessible nor are they appealing to replicate.
Our Understanding of the Diversity of Freshwater
These do not represent the diversity of freshwater ecosystems, even in the Brazilian Amazon these ideas do not stand (Bogotá-Gregory et al., 2020). There is frequent generalisation of countries and continents, how often do you hear that Africa is hard water? Yet even just the Rift Valley represents a diversity of ecosystems (Nyingi et al., 2013). This diversity of freshwater ecosystems is likely partially why over 50% of fishes are freshwater, if there was that homogenous nature across countries and continents we would see many more cosmopolitan general species.
Not just do we as a hobby maybe not understand how diverse freshwater is but how rivers function, source to mouth. It’s not just the hobby, there is a lack of research and understanding from the scientific community in certain areas of the world, particularly the tropics (Faghihinia et al., 2021). Much of us have not had an education in river geography, I myself took an optional module during undergraduate to get the basics. Yet marine biology is extensively taught. So we are left with maybe a school education on rivers which at least in the UK is terribly poor, additionally given most people come out of school only being able to say how an oxbow lake is formed.
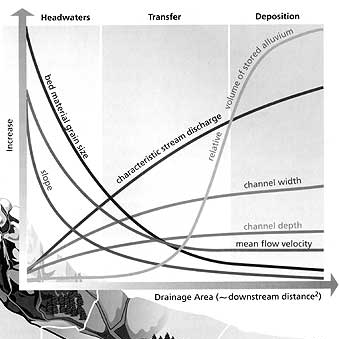
This article is not a lesson in how rivers function but Figure 1 is probably the most important graph in starting to understand how rivers function. It doesn’t just show function but shows diversity within the same water body, this should suggest even one river is not homogenous.
Blackwater
I should include this with a focus. Black water and botanicals, a growing popularity as people search for what is natural and what is not. As previously stated this style of fishkeeping is not representative of the diversity of freshwater ecosystems even within South America (Bogotá-Gregory et al., 2020). If we look at videos of rivers such as my favourite and a popular locality for Loricariids in the trade, the Rio Xingu there is little leaf litter in these localities opposed to largely biofilms If the reason for choosing black water is to replicate an ecosystem then I would argue against.

As a source of nutrients this is dependent on the water body in focus, even with high availability leaf litter is not always shown to be the source of energy opposed to algaes (Lewis et al., 2001). Any abiotic influences of botanicals particularly leaf litter is extremely diverse (Tonin et al., 2017). It’s not so simple basically.
I sound so very against botanicals but that is not my aim, my aim here is simply to say it is not always representative of tropical freshwater ecosystems. It has it’s place, a brilliant place, it provides enrichment and a way of replicating specific freshwater ecosystems e.g the Rio Negro. Of course there are other things to consider like oxygen saturation and waste removal in deep leaf litter, large amounts of nutrient introduction with certain botanicals e.g. palm leaves. The main point I’m making is, is it natural? Yes it is an attempt of replicating a freshwater ecosystem but is this the freshwater ecosystem of where the fish is found?
I think personally where black water has become increasingly possible many aquarists have taken it the wrong way.
The Planted Aquarium
Planted aquariums have been a staple of the aquarium hobby probably since the start. Ponds as well as a vital part of British horticulture and landscaping have long used plants. Our use of them has changed a lot, maybe from functional to aesthetic and then functional? I wont discuss the function here as that is a whole other discussion that is much more complex but I will say simply, they do not make an aquarium more stable.

I have mentioned earlier how plants might not be present or found in large numbers in freshwater habitats (Murphy et al., 2019). The presence and availability of plants in freshwater ecosystems is limited by factors linked often to stream order such as sedimentation, light availability, nutrient availability etc. (Fig 1). In freshwater where there might not be the sediment available for plants to root down you might see microbes such as photosynthetic bacteria, algaes and protozoa where they can cling on but take advantage of the light. There are other elements of freshwater which might be harsh to plants, the lack of light availability further downstream but there are plants who have evolved adaptation to this e.g. water lilies, Nymphaea growing fast to the surface or floating plants.
Where botanicals have been used from the influence of biotopes the planted aquarium is quite different. A few years ago natural was coined by aquascapers to describe a planted tank and now to be a tank with just plants but strongly influenced from the work of controversial Diana Walstad. There are so many variabilities with these new natural planted aquariums they are so difficult to discuss variabilities such as plant species, nutrient diversity and misunderstanding of freshwater ecosystems they are difficult to discuss.
I come from a horticulture background, my father who always had a diversity of exotic plants before moving to allotments and my mother a professional gardener, many of my grandparents and family have connections to horticulture. I grew up watching BBC Gardeners World. This has meant understanding of plants and their care is the norm to me, I’ve had house plants since my first memories and was encouraged to have a garden. This information about me is not entirely important, but from a young age it was ingrained the importance of different compounds depending on the species of plant, the pH of the soil and many abiotic factors. I am also a house plant enthusiast so keep relatives or the plants we often keep on our aquariums. Many of these plants are not being kept as how they would be found in the wild, this effects their morphology and their growth. Nutrient availability particularly ratios of each compound is important for many plants. Soil is not soil or dirt, soil is a substrate that represents many different types.

This understanding of soil I feel lacks the biggest understanding in the hobby as there is a wide diversity of different soils, even the reptile hobby recognises this. We have clay, sand, silt, loam, peat and chalk according to the Royal Horticulture Society (https://www.rhs.org.uk/soil-composts-mulches/soil-types) but more importantly the combination of which massively effects how plants might grow and which plants. In aquariums we also have to think about algaes and other microbes who will take advantage of this nutrients. Like earlier the amount of one compound/nutrients can effect the uptake of another. In horticulture we are only thinking about the plants but aquariums there are other organisms, even in horticulture when thinking about other organisms we can go drastically wrong.
I don’t think I can easily explain this whole topic in one article. Planted tanks are often more human-centric. Aquascapes often focusing on the human requirements of aesthetics, maybe lacking hiding spaces or where a fish might need to dig, definitely worth thinking about. In comparison the recent popular planted aquariums don’t focus on the fishes, never mentioning fish biology.
Why do I discuss this?
I am a fishkeeper and a biologist but freshwater fishes are my passion. Of all the aspects of the hobby the word natural has been misused as a keyword. Maybe nothing I say here matters because it is used as clickbait more then often. What matters more is to research individual fishes and their biology. If you want to keep plants, keep plants but consider the fishes if you’re adding them.
My passion is largely Loricariids and rasping fishes, regarding black water and botanicals there are definitely a large number of Loricariids found in ecosystems with those, mostly representatives of the subfamilies Loricariinae and Hypoptopominae. These planted ‘natural’ aquariums often do not provide enough current, not enough water changes and not enough oxygen, temperature. This is likely due to Loricariids being seen as generalist even if that is a total myth.
References:
Bogotá-Gregory, J. D., Lima, F. C., Correa, S. B., Silva-Oliveira, C., Jenkins, D. G., Ribeiro, F. R., … & Crampton, W. G. (2020). Biogeochemical water type influences community composition, species richness, and biomass in megadiverse Amazonian fish assemblages. Scientific Reports, 10(1), 15349.
Department for Environmental and Rural Affairs, Forestry England, Animal and Plant Health Agency & Gardiner, J. (2019). Invasive Species Week: American skunk cabbage creates a stink. GOV.UK. https://www.gov.uk/government/news/invasive-species-week-american-skunk-cabbage-creates-a-stink#:~:text=American%20skunk%20cabbage%20was%20first,an%20ornamental%20plant%20in%201901.
Eastwood, N., Zhou, J., Derelle, R., Abdallah, M. A. E., Stubbings, W. A., Jia, Y., … & Orsini, L. (2023). 100 years of anthropogenic impact causes changes in freshwater functional biodiversity. bioRxiv, 2023-02.
Faghihinia, M., Xu, Y., Liu, D., & Wu, N. (2021). Freshwater biodiversity at different habitats: Research hotspots with persistent and emerging themes. Ecological Indicators, 129, 107926.
Franco, L. S., Shanahan, D. F., & Fuller, R. A. (2017). A review of the benefits of nature experiences: More than meets the eye. International journal of environmental research and public health, 14(8), 864.
Lewis Jr, W. M., Hamilton, S. K., Rodríguez, M. A., Saunders III, J. F., & Lasi, M. A. (2001). Foodweb analysis of the Orinoco floodplain based on production estimates and stable isotope data. Journal of the North American Benthological Society, 20(2), 241-254.
Murphy, K., Efremov, A., Davidson, T. A., Molina-Navarro, E., Fidanza, K., Betiol, T. C. C., … & Urrutia-Estrada, J. (2019). World distribution, diversity and endemism of aquatic macrophytes. Aquatic Botany, 158, 103127.
Nyingi, D. W., Gichuki, N., & Ogada, M. O. (2013). Freshwater ecology of Kenyan highlands and lowlands. In Developments in earth surface processes (Vol. 16, pp. 199-218). Elsevier.
Schultz, R., & Dibble, E. (2012). Effects of invasive macrophytes on freshwater fish and macroinvertebrate communities: the role of invasive plant traits. Hydrobiologia, 684, 1-14.
Scott, S. E., Rozin, P., & Small, D. A. (2020). Consumers prefer “natural” more for preventatives than for curatives. Journal of Consumer Research, 47(3), 454-471.
Søndergaard, M., & Jeppesen, E. (2007). Anthropogenic impacts on lake and stream ecosystems, and approaches to restoration. Journal of applied ecology, 44(6), 1089-1094.
Simpson, D. A. (1986). Taxonomy of Elodea Michx in the British Isles. Watsonia, 16, 1-14.
Tonin, A. M., Goncalves Jr, J. F., Bambi, P., Couceiro, S. R., Feitoza, L. A., Fontana, L. E., … & Boyero, L. (2017). Plant litter dynamics in the forest-stream interface: precipitation is a major control across tropical biomes. Scientific Reports, 7(1), 10799.
Wilson, E. O. (2007). Biophilia and the conservation ethic. Evolutionary perspectives on environmental problems, 249-257.